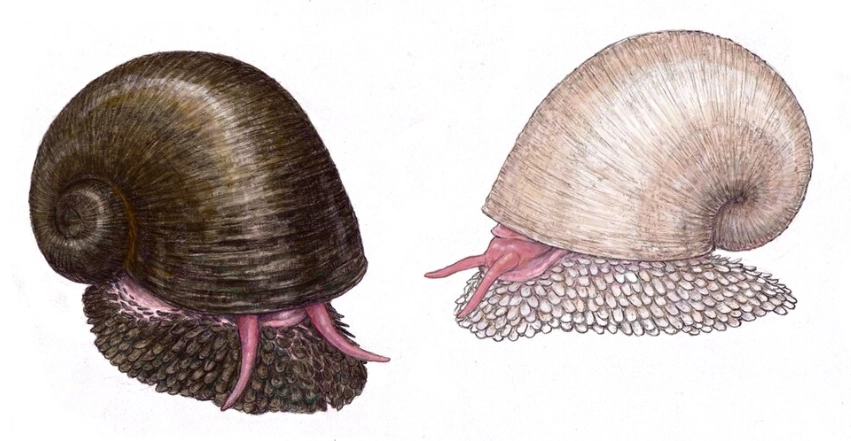
Let's cut straight to the chase. Yes, the scaly-foot snail is absolutely real. It's not a Photoshop creation, a mythical sea monster, or a prop from a sci-fi movie. Chrysomallon squamiferum is a genuine, living, breathing (well, gill-breathing) snail that calls one of the most extreme environments on Earth its home. If you stumbled upon a picture and thought, "No way that's real," you're in good company. Its appearance is so outlandish it feels like it should be fake. But the reality is far more fascinating than any fiction.
I remember the first time I saw a research photo. It looked like a tiny, discarded knight's gauntlet made of iron, sitting on a black smoker vent. The sheer improbability of it hooked me. This creature doesn't just exist; it thrives under conditions that would instantly kill almost every other animal on the planet. But its "cool factor" is shadowed by a pressing reality: it was the first species to be declared endangered due to the threat of deep-sea mining, even before mining had officially begun. That's a sobering headline.
What You'll Find in This Deep Dive
The Scaly-Foot Snail at a Glance
- Scientific Name: Chrysomallon squamiferum
- Nicknames: Iron snail, Sea pangolin, Scaly-foot gastropod
- Home: Deep-sea hydrothermal vents in the Indian Ocean
- Depth: About 2,400–2,900 meters (7,900–9,500 feet) down
- Key Feature: Its foot is covered in sclerites—mineralized scales containing iron sulfides.
- Conservation Status: Endangered (IUCN Red List)
- Primary Threat: Potential deep-sea mining for polymetallic sulfides.
What Exactly Is the Scaly-Foot Snail?
Forget everything you know about garden snails. Chrysomallon squamiferum is an extremophile, an organism that lives in conditions of extreme temperature, pressure, and chemistry. It's a gastropod, yes, but it's evolved solutions to problems most life never encounters.
Its most famous feature is its scaly foot. These aren't like fish scales; they're called sclerites. They're like little armored plates made of organic material and minerals, primarily iron sulfides (greigite and pyrite) – that's fool's gold. No other animal on Earth is known to incorporate iron sulfides into its skeletal structure. The snail's shell is also unique, with an organic middle layer that acts as a shock absorber, a brilliant adaptation to withstand the bumps and knocks of life near turbulent vent chimneys.
How Was It Discovered?
The story of its discovery is a classic tale of deep-sea exploration. It wasn't found by accident on some beach. The scaly-foot snail was first collected in 2001 during a Japanese research cruise to the Kairei hydrothermal vent field on the Central Indian Ridge. Scientists piloting the submersible Shinkai 6500 spotted these bizarre, metallic-looking snails clustered around the superheated, mineral-rich fluids spewing from the seafloor.
The initial scientific description was published in 2003, and it immediately turned heads in the marine biology community. Here was an animal that seemed to defy basic biological principles. It wasn't just surviving in a toxic environment; it was actively using the toxins (like hydrogen sulfide) and metals to build its own body armor. It was a living lesson in bio-mineralization.
Why Is It So Unique? The Iron Snail Breakdown
Let's break down what makes this snail a biological superstar. Most articles will list its iron scales and call it a day. But the real magic is in the why and how.
1. The Iron Sulfide Armor (The "Scaly Foot")
The sclerites aren't just for show. Research suggests they serve multiple critical functions:
Physical Defense: They create a tough, abrasive barrier against predators. In the deep sea, a major threat is attack from crabs and other crustaceans. Try pinching through iron-reinforced scales.
Thermal Insulation: The vent environment is a patchwork of extremes—scalding hot water (over 300°C/572°F) from the vents and near-freezing ambient seawater. The mineral composition of the sclerites may help manage this thermal shock.
Chemical Detoxification: This is a subtle point many miss. The vent fluids are rich in hydrogen sulfide, which is poisonous to most animals because it blocks cellular respiration. A leading theory is that the snail's symbiotic bacteria (living in a special organ) use the sulfide as an energy source. The iron in the sclerites may help sequester excess sulfide, preventing it from poisoning the snail itself. It's like having built-in, reactive body armor.
2. The Three-Layer Shell
Its shell is a masterpiece of engineering. Unlike a typical snail shell, it has three distinct layers:
- Outer layer: Made of iron sulfides, similar to the sclerites.
- Middle layer: A thick, organic, rubber-like layer of conchiolin. This is the genius part—it acts as a shock absorber, dissipating the energy from a crab's claw or a rock fall. It's nature's version of bulletproof vest padding.
- Inner layer: A classic aragonite (calcium carbonate) layer, like in many other mollusks.
3. It Farms Its Own Food (Symbiosis)
The scaly-foot snail doesn't graze on algae or detritus. There's no sunlight for algae 2.5 kilometers down. Instead, it has a massive esophageal gland that houses chemosynthetic bacteria. These bacteria use the hydrogen sulfide from the vent fluids as an energy source to produce organic carbon—food for the snail. The snail provides the bacteria with a safe home and access to chemicals. It's a perfect, closed-loop farming system in one of the most barren environments on the planet.
Where Does It Live and What's Killing It?
This is where the story turns from wonder to worry. The scaly-foot snail has an incredibly limited distribution. As far as we know, it only lives in three hydrothermal vent fields in the Indian Ocean:
- The Kairei Field (Central Indian Ridge, where it was first found).
- The Longqi Field (also known as Dragon's Breath, Southwest Indian Ridge).
- The Solitaire Field (Central Indian Ridge).
These vents are like isolated, deep-sea islands. The snails at one vent field cannot travel to another. They are trapped in their own tiny, extreme worlds.
The threat? The very thing that makes its home unique: the mineral deposits. Hydrothermal vents are rich in copper, zinc, gold, silver, and rare earth elements. These are polymetallic sulfides, and they are the target of emerging deep-sea mining ventures. A single mining operation could completely obliterate an entire vent field—and with it, the entire population of scaly-foot snails living there.
This is why the International Union for Conservation of Nature (IUCN) listed it as Endangered in 2019. It was a landmark decision—assessing a species as threatened by a commercial activity that hadn't even started yet, based on the foreseeable risk. It's a precautionary tale for the entire deep-sea ecosystem.
Some argue that deep-sea mining is necessary for the green energy transition (to get metals for batteries). But wiping out entire ecosystems we've barely begun to understand, and creatures as unique as the iron snail, seems like a catastrophic trade-off. We might destroy potential medical or engineering breakthroughs (like new, super-strong materials inspired by its shell) before we even discover them.
Your Top Questions About the Iron Snail
 Can I see a scaly-foot snail in an aquarium?
Can I see a scaly-foot snail in an aquarium?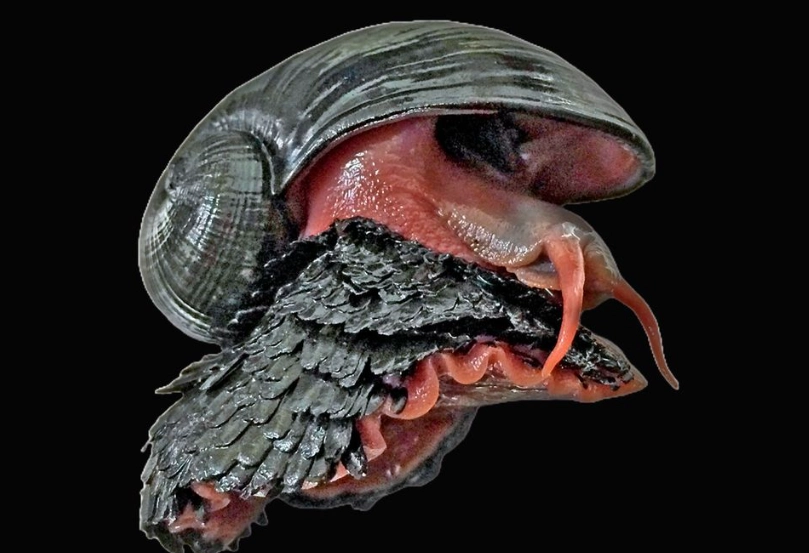 What's being done to protect it?
What's being done to protect it?So, is the scaly-foot snail real? More than real. It's a testament to life's incredible adaptability, a piece of living engineering that humbles our own technology, and a stark warning about the hidden costs of resource extraction. It exists in a fragile, dark world at the bottom of the sea, a world we are just beginning to explore but are already poised to destroy. Understanding this creature isn't just about satisfying curiosity; it's about recognizing the value of the unknown and the irreplaceable wonders we stand to lose.






Comments
Leave a Comment