Let's get one thing straight right away: the scaly-foot gastropod (Chrysomallon squamiferum) isn't your average garden snail. Forget the delicate, chalky shell you might be picturing. This creature, found only in the crushing darkness of deep-sea hydrothermal vents, builds its home out of iron. That's right. Its shell and even the scales on its foot are reinforced with iron sulfides, making it the only known animal on Earth to incorporate metallic elements into its skeleton. It's less a snail and more a living, breathing suit of armor, forged in one of the planet's most extreme environments.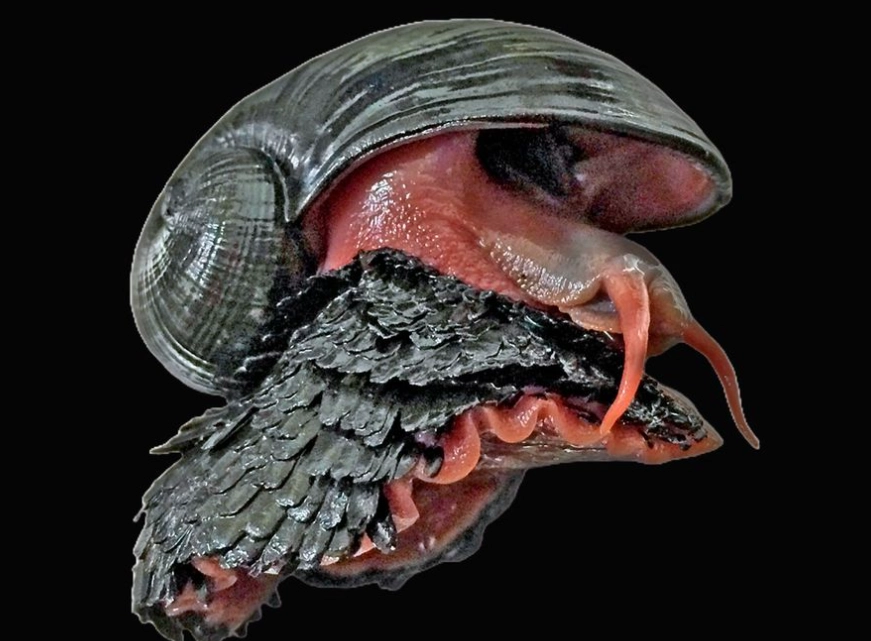
Dive into the Iron Snail’s World
What Exactly Is the Scaly-foot Gastropod?
The scaly-foot gastropod is a species of deep-sea snail discovered in 2001 near the Kairei hydrothermal vent field in the Indian Ocean. It belongs to a family of vent snails, but it stands utterly alone in its construction methods. The entire animal is an adaptation to a brutal world of superheated, mineral-rich water, toxic chemicals, and immense pressure.
Its most famous feature is, of course, its shell. But the name "scaly-foot" comes from the hundreds of hard, overlapping scales covering its fleshy foot—the part it uses to move. Both the shell and these scales are mineralized with greigite and pyrite, which are iron sulfides. In simple terms, it's wearing iron plates. This isn't just for show. In the vent environment, this armor serves as a defense against predators like crabs, whose powerful claws would make quick work of a normal calcium carbonate shell.
Where Does This Iron Snail Live? The Hunt for Hydrothermal Vents
You can't just go snorkeling to find one. The scaly-foot gastropod is an extremophile, thriving in conditions that would instantly kill most life. Its entire world is a few, highly specific locations on the ocean floor.
Its primary known habitats are three hydrothermal vent fields along the Central Indian Ridge:
- The Kairei Vent Field: The original discovery site, about 2,420 meters deep. This is the "type locality" for the species.
- The Longqi Vent Field (Dragon's Breath): Located further southwest, at a depth of around 2,800 meters.
- The Solitaire Field: Another site where populations have been confirmed.
Finding these vents is a monumental task requiring specialized research vessels like the RV Yokosuka and deep-diving submersibles like Shinkai 6500. The "address" is essentially GPS coordinates over 2 km below the surface, near geological formations where tectonic plates are pulling apart. The "business hours" are 24/7, in perpetual darkness, with water temperatures around the vents swinging from near freezing to over 300°C (572°F). The journey there is more akin to a space mission than a beach trip.
This extreme localization is a double-edged sword. It makes the snail a fascinating subject for study, but it also makes the entire species incredibly vulnerable. A single event, like a subsea mining operation or a major geological shift, could wipe out a significant portion of its global population.
A Breakdown of Its Incredible Three-Layer Armor
Here’s where the scaly-foot gastropod shell moves from cool fact to engineering masterpiece. It's not a solid lump of iron. Research, notably from scientists at MIT and Harvard who have published papers in journals like Proceedings of the National Academy of Sciences, reveals it's a sophisticated, composite material with three distinct layers, each with a specific job.
I remember reading one of the early papers and being stunned by the elegance of it. It’s not brute force; it’s clever design.
| Layer | Material Composition | Primary Function | Analogy |
|---|---|---|---|
| Outer Layer | Iron sulfides (greigite granules) | Impact resistance; sacrificial wear layer. Hardens and becomes more abrasion-resistant over time. | The hardened steel plating on a tank. It takes the direct hits and scuffs. |
| Middle Layer | Soft, organic periostracum (a protein matrix) | Energy dissipation. Acts as a flexible, squishy pad that absorbs shock and prevents cracks from propagating inward. | The rubbery padding inside a football helmet. It soaks up the force. |
| Inner Layer | Calcium carbonate (aragonite) | Structural support and protection against chemical corrosion from the snail's own acidic body fluids. | The reinforced concrete inner wall. It provides the core shape and protects from the inside out. |
This layered defense is key. A common misconception is that the iron makes the shell indestructible. It doesn't. The genius is in the combination. The hard outer layer resists the initial puncture or crush. If a crack starts, it hits the soft middle layer, which blunts it and spreads the energy out. The inner layer remains intact, protecting the soft animal inside. It's a perfect system for resisting the powerful, pinpoint attacks of a crab's claw.
The scales on its foot operate on a similar principle, creating a flexible yet formidable mail coat. Frankly, it looks a bit... clunky. But in its high-pressure, predator-filled world, fashion takes a back seat to function.
Beyond the Wow Factor: Why This Snail Matters to Science
So we have a cool snail with an iron suit. Why should anyone but a handful of marine biologists care? The implications stretch far beyond the deep sea.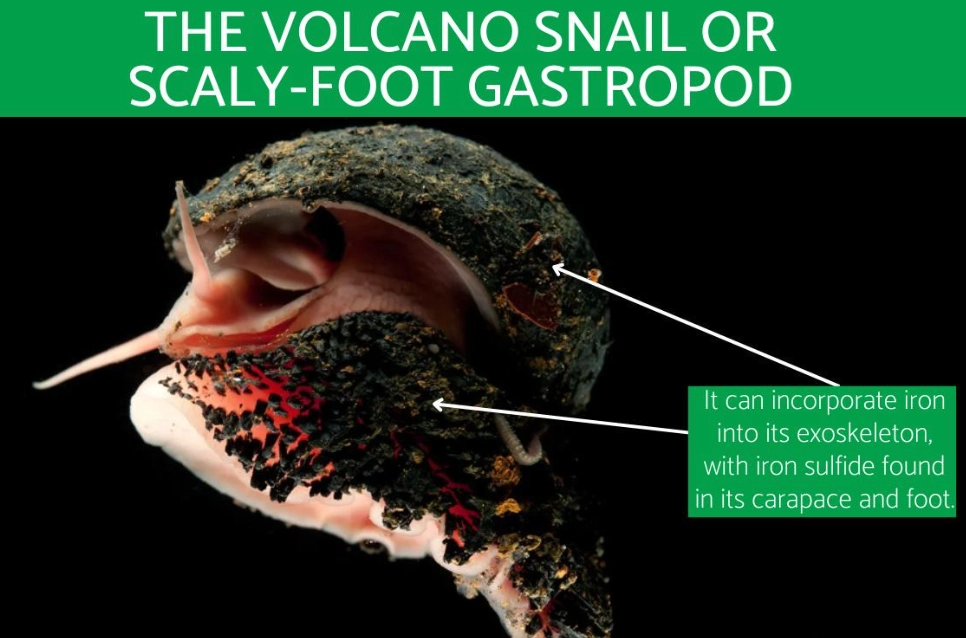
First, it's a masterclass in evolution. This snail showcases how life can innovate with locally available materials. Instead of relying solely on calcium carbonate, which is the standard issue for mollusks, it incorporated the abundant iron and sulfur spewing from the vents. It's a real-time example of extreme adaptation.
Second, and perhaps most importantly, it's a goldmine for biomimicry and materials science. Engineers are desperately trying to design better lightweight armor, impact-resistant coatings, and durable composites. The scaly-foot gastropod's shell provides a blueprints. That three-layer structure—a hard exterior, a dissipative middle, and a tough interior—is a design principle that could revolutionize everything from body armor for soldiers to protective casings for smartphones and drones. Research institutions like the Woods Hole Oceanographic Institution often highlight such deep-sea discoveries for their potential technological spin-offs.
Finally, it's a canary in the coal mine for deep-sea ecosystems. Its existence is tied to the fragile and rare hydrothermal vent ecosystems. Studying its health and population helps us understand the impacts of deep-sea mining and climate change on these isolated oases of life.
Threats and the Uncertain Future of a Deep-Sea Icon
This brings us to the sobering part. The scaly-foot gastropod was classified as Endangered on the IUCN Red List in 2019. Its future is precarious.
The main threat isn't overfishing or pollution in the traditional sense. It's the potential for deep-sea mining. The very hydrothermal vents it calls home are rich in valuable metals like copper, zinc, gold, and silver. Mining companies are eyeing these resources. The process is devastating: massive machines would crawl across the seafloor, scraping up the mineral-rich chimneys and the surrounding substrate, utterly destroying the habitat. For a snail that lives only in these specific spots and moves very slowly, this is an existential threat. There's no "next vent over" to migrate to.
Other threats include scientific collection (though tightly regulated) and the potential long-term effects of climate change on deep ocean currents and chemistry. Conservation efforts are complex because they involve international waters and balancing economic interests with biodiversity protection. Organizations like the International Union for Conservation of Nature (IUCN) are advocating for strong regulations and marine protected areas around vulnerable vent sites before mining begins.
Your Iron Snail Questions Answered
In an environment like a hydrothermal vent where crabs are a major predator, is the iron shell really more advantageous than a traditional calcium carbonate one?
Absolutely, but not because iron is inherently "stronger" in a simple sense. Calcium carbonate shells can be very strong, but they are brittle. A crab's claw applies immense, focused pressure—it's a crack-propagation machine. The iron sulfide outer layer is harder and more resistant to that initial puncture and abrasion. More crucially, the composite layered structure (hard/soft/tough) is uniquely suited to dissipating the energy of a pinch or crush, preventing a single crack from shattering the entire shell. A pure calcium carbonate shell in that environment would be a liability.
I've read that the snail gets its iron from bacteria. Is that true, and how does that process work?
This is a subtle point that's often oversimplified. The snail doesn't "eat" iron from the bacteria. Here's the real process: The snail hosts chemosynthetic bacteria in a special organ. These bacteria use hydrogen sulfide from the vent fluids as an energy source to produce organic carbon (food for the snail). The dissolved iron (from the vent fluids) and sulfur are likely absorbed directly from the water by the snail's tissues. The snail's own cells then control the biomineralization process, secreting the iron sulfide nanoparticles to form the shell and scales. The bacteria are the energy providers, not the iron delivery service. The snail is the brilliant architect and builder.
 Could we ever see scaly-foot gastropods in public aquariums?
Could we ever see scaly-foot gastropods in public aquariums?
It's extremely unlikely, and that's a good thing for the snail. The technical challenges are immense. You'd need to replicate the extreme pressure (over 240 atmospheres), the specific chemical soup of the vent water (high in sulfides, metals, with zero oxygen), and maintain a stable temperature gradient—all in a closed system. The cost and energy required would be astronomical. More importantly, collecting enough individuals to establish a breeding population could harm the wild colonies. Some things are better left as wonders of the deep, studied remotely by ROVs. Their mystique is part of their magic.
If the shell is so good, why aren't there more animals with iron armor?
Evolution is about trade-offs. Iron sulfides are heavy and metabolically expensive to produce. They also require a specific environment rich in dissolved iron and sulfur, which is rare on Earth's surface. For most animals, the benefits don't outweigh the costs. Calcium carbonate is lighter, easier to produce from readily available materials (calcium and carbonate in seawater), and perfectly adequate for most ecological niches. The scaly-foot gastropod evolved in a unique, metal-rich, high-threat environment where the heavy investment in iron armor paid off. It's a specialist's solution, not a universal one.






Comments
Leave a Comment